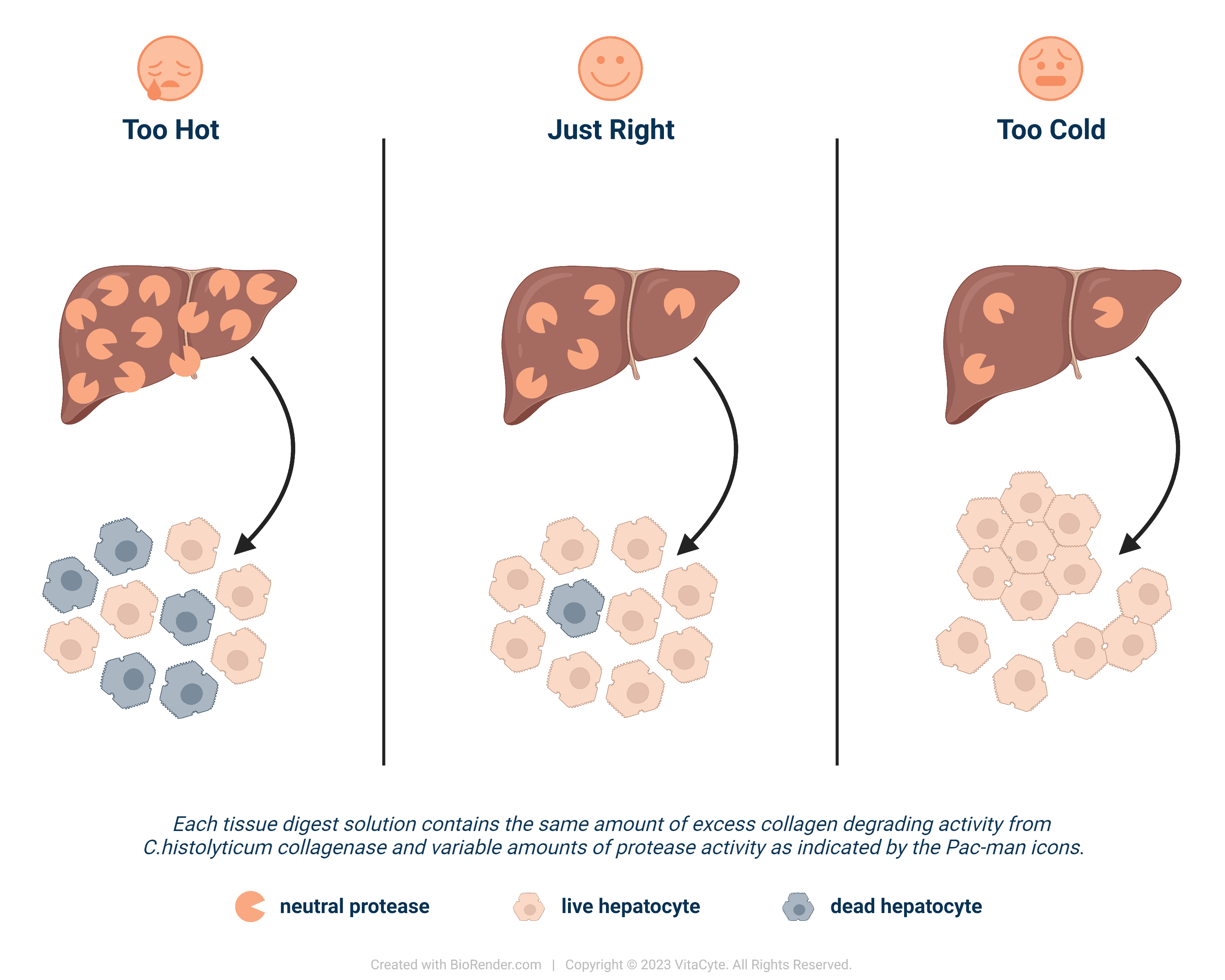
Astronomers search for life on other planets coined the phrase the “Goldilocks Zone” to describe the ideal conditions that will support life. The “life supporting” planet cannot be too close or too far away from the sun of its solar system. It must have temperate zones that allows liquid water to exist and have an inhabitable atmosphere that supports the life forms.
This concept can be extended to describe the balance of collagenase and protease enzymes used to obtain optimal yields of functional cells after enzyme mediated cell isolation. Here, the conditions for optimal cell isolation are summarized in five postulates:
- The extracellular matrix (ECM) is resistant to proteolysis with the collagen fibrils/fibers providing the superstructure to protect other ECM proteins from protease degradation, minimizing damage to the cellular architecture of the tissue.
- Purified C. histolyticum collagenase has a narrow specificity, degrading only gelatin and native collagen in the extracellular matrix, excess amounts of purified collagenase will not damage cells.
- Class I and class II collagenase containing a catalytic domain and at least one collagen binding domain are the only functional forms of the enzyme that can degrade native collagen.
- To ensure cell release, C. histolyticum collagenases’ collagen degradation activity (CDA) must be in excess to loosen the ECM, leading to exposure of protease sensitive sites on other ECM proteins.
- Once a sufficient number of “cell anchoring ECM proteins” cut, cells released from tissue.
The “just right” Goldilocks’ Zone is achieved when the environment contains excess amount of collagen degrading activity from purified C. histolyticum collagenase is present in the enzyme solution used for tissue digestion. The amount of neutral protease activity from purified enzymes is the determining factor for achieving “just right” conditions. “Too hot” (high protease) will damage cells leading to lower cell viabilities. “Too cold” occurs when an insufficient amount of protease is unable to cut all of the cell anchoring proteins. This results in many clumped cells. In “just right”, an optimal number of viable, functional cells are recovered.
Why use purified enzymes to optimize cell isolation conditions? Why can’t the same conditions apply to traditional crude and enriched collagenase products? The fundamental limitation for optimizing traditional collagenase products for cell isolation is the ill-defined nature of the collagenase and protease present in a product that contains anywhere from 3-8% (w/w) and 10-20% collagenase for crude and enriched collagenase, respectively. The CDA measurements are imprecise, using a poorly characterized research assay developed in 1953. There is no knowledge of the different molecular forms of functional collagenase in these products. Moreover, variable amounts of neutral protease activities contributed by two enzymes: C. histolyticum neutral protease and clostripain are present in each lot of product. If a high amount clostripain present, it can degrade class I collagenase as it is stored as a lyophilized powder or frozen liquid.
The above explains why many cell isolators perform lot qualification of collagenase before purchasing a large amount for routine use. The method relies on the expectation that at least one of the lots tested will perform similarly to the current lot of collagenase product. The enzyme components found in a “good lot” of product likely falls into the Goldilocks’ Zone. However, once that lot is depleted, you fall into another scenario, “Groundhog Day,” where you replay your collagenase lot qualification process often annually. Why support unproductive, non-value adding work? It is your choice.
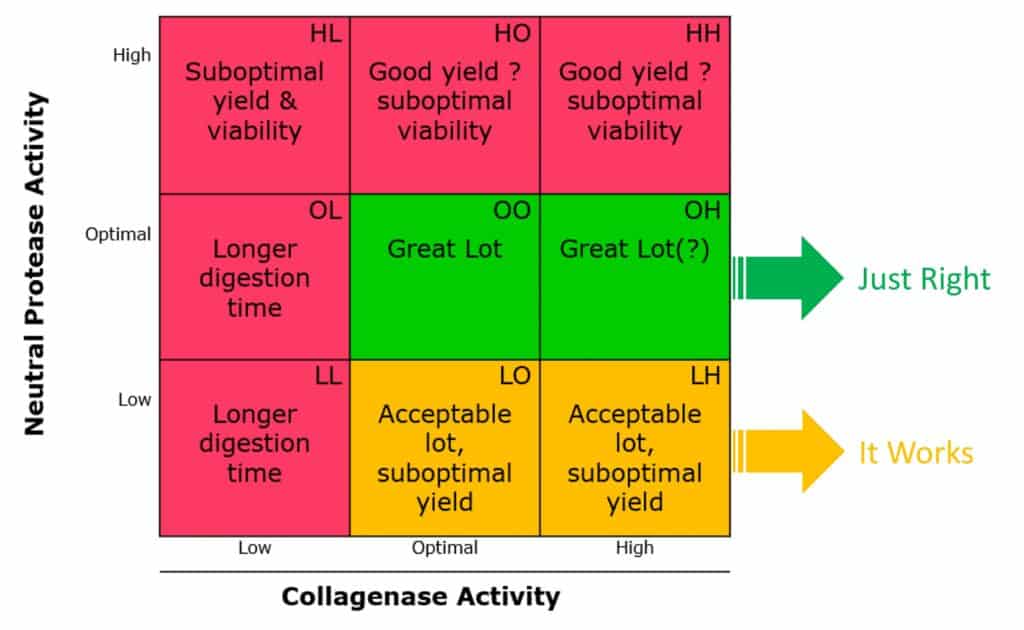
The concepts above are summarized in the “enzyme-mediated cell isolation matrix” below. The best approach to control collagenase-mediated cell isolation processes is to use purified, defined enzymes where the cell isolator controls the enzyme composition and not the other way around, where you hope to obtain a lot of crude or enriched collagenase products that perform similarly to your current lot of collagenase.